An Innovative Class of Dialyzer Membrane Technology
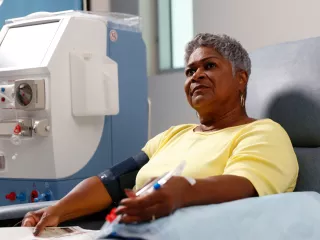
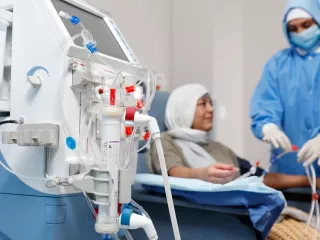
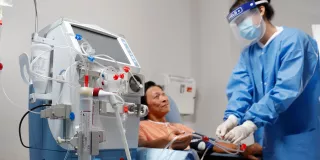
As the hemodialyzer with expanded solute removal profile, Theranova dialyzer and its MCO membrane enable HDx therapy to provide expanded hemodialysis.1
Renal care resources to support clinicians
We offer numerous resources that provide detailed information on using our AK 98 dialysis system, which can deliver HDx therapy using Theranova dialyzer, and on our support services for both clinicians and patients.
Important Safety Information
Indications for Use: THERANOVA dialyzers are indicated for treatment of chronic and acute renal failure by Hemodialysis
Caution: Do not use THERANOVA dialyzers for HDF (hemodiafiltration) or HF (hemofiltration) due to higher permeability of larger molecular weight proteins such as albumin.
For safe and proper use of the device, please refer to the Instructions for Use.
Vantive, AK 98, HDx, MCO and Theranova are trademarks of Vantive Health LLC or its affiliates.
References
-
Device Classification Under Section 513(f)(2)(DeNovo). Accessdata.fda.gov. https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/denovo.cfm?id=DEN190042. Published 2020.
-
Zweigart C, Boschetti-de-Fierro A, Hulko M, et al. Medium cut-off membranes — closer to the natural kidney removal function. Int J Artif Organs. 2017;40(7):328-334.
-
Ronco C, La Manna G. Expanded hemodialysis: a new therapy for a new class of membranes. Contrib Nephrol. 2017;190:124-133.
-
Ronco C. The rise of expanded hemodialysis. Blood Purif. 2017;44(2):I-VIII.