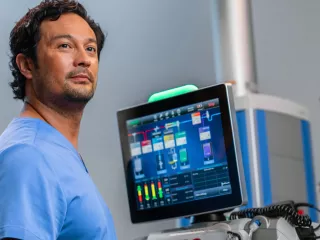
Partnering on the Front Line of Intensive Care
Vantive’s acute therapy systems help clinicians individualize organ support therapy for their patients, while streamlining the complexities of critical care.
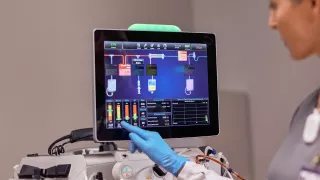
Prismax System
Our continuously evolving PrisMax System is designed to help clinicians optimize therapy delivery and quality of care for critically ill patients by maximizing simplicity, efficiency and accuracy in CRRT.

PrisMax 2 System
Deliver a range of therapies on the PrisMax 2 System – CRRT, TPE and blood purification for sepsis management to remove cytokines and endotoxins – without adding complexities in delivery.

Prismaflex System
For continuous renal replacement therapy (CRRT), the Prismaflex System supports the individual needs of patients with single or multiple organ failure therapies in one device.
PrisMax 2 System | PrisMax System | Prismaflex System | |
CRRT Filtersets | ✓ | ✓ | ✓ |
Oxiris Set | ✓ | ✓ | ✓ |
TPE Filtration Sets | ✓ | ✓ | ✓ |
GamCath Short-Term Catheters | ✓ | ✓ | ✓ |
TrueVue | ✓ | ✓ | ⤫ |
Important Safety Information
For safe and proper use of the devices, please refer to the Instructions for Use.
PrisMax:
- The PrisMax control unit is intended for:
Continuous Renal Replacement Therapy (CRRT) for patients with acute renal failure and/or fluid overload.
- Therapeutic Plasma Exchange (TPE) therapy for patients with diseases where removal of plasma components is indicated.
- Hemoperfusion (HP) for patients with conditions where immediate removal of substances by adsorption is indicated.
All treatments administered via the PrisMax control unit must be prescribed by a physician.
WARNING!
Use only fluids designed for intravenous operations and are approved for CRRT/TPE/HP usage. Do not use fluids intended for any other usage like enteral feedings/lipids or cytotoxic drugs.
CONTRAINDICATIONS
There are no known absolute contraindications to Continuous Renal Replacement Therapies.
There are no known contraindications to Therapeutic Plasma Exchange.
There are no known contraindications to Hemoperfusion.
Regional Citrate Anticoagulation is contraindicated in patients with severe liver failure and shock with muscle hypoperfusion.
For the following conditions a careful assessment of the individual risk/benefit ratio has to be made by the treating physician (relative contraindications).
- Inability to establish vascular access.
- Severe hemodynamic instability.
- Known hypersensitivity to any component of the Prismaflex disposable set.
PRISMAFLEX
Intended use
The PRISMAFLEX control unit is intended for:
- Continuous Renal Replacement Therapy for patients with acute renal failure and/or fluid overload.
- Therapeutic Plasma Exchange Therapy for patients with diseases where removal of plasma components is indicated.
- Hemoperfusion for patients with conditions where immediate removal of substances by adsorption is indicated.
All treatments administered via the PRISMAFLEX control unit must be prescribed by a physician.
Contraindications
There are no known contraindications to Continuous Renal Replacement Therapies.
There are no known contraindications to Therapeutic Plasma Exchange.
There are no known contraindications to Hemoperfusion.
For contraindications that may apply to the disposable set selected for the therapy, refer to the Instructions For Use of the disposable set.
CE 0123
CE 2797
Vantive, GamCath, Oxiris, Prismaflex, PrisMax and TrueVue are trademarks of Vantive Health LLC or its affiliates.