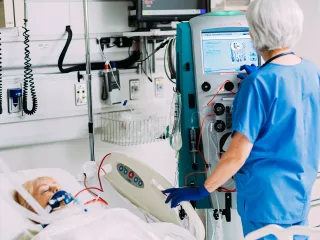
Prismaflex System
One device for single or multiple organ failure support
One of the world’s most used CRRT devices
When treating critically ill patients, providers need a device they can rely on. For continuous renal replacement therapy (CRRT), the Prismaflex System supports the individual needs of patients with single or multiple organ failure therapies in one device. Prismaflex is a versatile machine with a comprehensive product offering, along with training and support to help deliver optimized outcomes for the ICU.
Compatible Products
Acute Therapy Systems
The Prismaflex System continues to advance therapy delivery as one of the world’s most used CRRT devices, with thousands of systems installed in over 90 countries.
Oxiris Set
The only 3-in-1 blood purification filterset to effectively remove inflammatory mediators such as cytokines and endotoxins as a blood purification therapy for sepsis management. Therapy can be delivered as a stand alone hemoperfusion treatment or while delivering CRRT.2
Integrated Citrate and Calcium Management
Fully integrated citrate and calcium management to enable delivery of regional citrate anticoagulation for all CRRT modalities.
AN69ST Membrane
Proprietary membrane that provides a unique combination of diffusion, convection and adsorption functionalities, which is clinically proven to provide removal of middle-size molecules in convective therapies3 or via adsorption.4,5
TPE Sets
Plasma protein removal in both adults and children (≥9 kg) for therapeutic plasma exchange (TPE) blood purification therapy.6
Smooth transition to the PrisMax 2 System
Busy clinicians must limit potential disruptions in workflow and nurse training that can come with switching to a new system. The PrisMax 2 System is built on the workflow Prismaflex users already know and comes with Vantive service, training and support to allow a smooth transition from Prismaflex to PrisMax 2.
- Familiar therapies and modalities
- Continued use of same filtersets and solutions
- Similar alarm management
Important Safety Information
PrisMax:
- The PrisMax control unit is intended for:
Continuous Renal Replacement Therapy (CRRT) for patients with acute renal failure and/or fluid overload.
- Therapeutic Plasma Exchange (TPE) therapy for patients with diseases where removal of plasma components is indicated.
- Hemoperfusion (HP) for patients with conditions where immediate removal of substances by adsorption is indicated.
All treatments administered via the PrisMax control unit must be prescribed by a physician.
WARNING!
Use only fluids designed for intravenous operations and are approved for CRRT/TPE/HP usage. Do not use fluids intended for any other usage like enteral feedings/lipids or cytotoxic drugs.
CONTRAINDICATIONS
There are no known absolute contraindications to Continuous Renal Replacement Therapies.
There are no known contraindications to Therapeutic Plasma Exchange.
There are no known contraindications to Hemoperfusion.
Regional Citrate Anticoagulation is contraindicated in patients with severe liver failure and shock with muscle hypoperfusion.
For the following conditions a careful assessment of the individual risk/benefit ratio has to be made by the treating physician (relative contraindications).
- Inability to establish vascular access.
- Severe hemodynamic instability.
- Known hypersensitivity to any component of the Prismaflex disposable set.
TPE Sets:
The Prismaflex TPE1000/TPE2000 set is indicated for use only with the PrismaFlex control unit or with the PrisMax control unit (in countries where PrisMax is cleared or registered), after having selected TPE therapy (therapeutic plasma exchange).
The Prismaflex TPE1000/TPE2000 set is intended for use in therapeutic plasma exchange, thus in diseases where removal of plasma components in indicated.
The use of the Prismaflex TPE1000 set should be restricted to patients with a body weight greater than 9kgs (20lbs).
The use of the Prismaflex TPE2000 set should be restricted to adults.
The size, weight, state of uremia, cardiac status and general physical condition of the patient must be evaluated by the prescribing physician before each treatment.
The device should be used only on the direction of a physician who has evaluated all of the pertinent features of this device in relation to the individual patient. Patients with bleeding tendencies must be closely supervised during the treatment.
Treatment should be discontinued if acute hemorrhage that can not be corrected (hematemesis, hemoptysis and melena) occurs during TPE.
All treatments administered via the Prismaflex set must be prescribed by a physician.
Oxiris:
Intended Purpose
The oXiris set is a single use device that provides blood purification via diffusion, convection and adsorption through a semipermeable membrane.
The oXiris set is for use only in conjunction with the PrismaFlex control unit or with the PrisMax control unit (in countries where PrisMax is cleared or registered).
All treatments administered with the oXiris set must be prescribed by a physician. The size, weight, state of uremia, cardiac status, and general physical condition of the patient must be carefully evaluated by the prescribing physician before each treatment.
Indications
The oXiris set is indicated for use in critically ill patients with a body weight equal or greater than 30 kg (66lb) for hemoperfusion and / or renal replacement modalities such as:
- Slow Continuous UltraFiltration (SCUF)
- Continuous Veno-Venous Hemofiltration (CVVH)
- Continuous Veno-Venous HemoDialysis (CVVHD)
- Continuous Veno-Venous HemoDiaFiltration (CVVHDF)
When used for hemoperfusion only, the SCUF mode shall be used with no fluid removal prescription, as the indication is to reduce elevated levels of inflammatory mediators, such as cytokines, and to reduce endotoxins.
If patients suffer from acute kidney injury and / or volume overload, the oXiris set is indicated for continuous renal replacement therapies (CRRT), to perform fluid management and removal of uremic toxins. The removal of inflammatory mediators and endotoxins is performed simultaneously when indicated for CRRT.
Contraindications
It is contraindicated to use the oXiris set where patients present a known allergy to heparin or have type II thrombocytopenia caused by heparin (HIT Syndrome type II).
Any contraindications indicated in the respective Instructions for use of all drugs used simultaneously with this set should be taken into account.
For the following conditions a careful assessment of the individual risk/benefit ratio has to be made by the treating physician (relative contraindications):
- inability to establish vascular access,
- severe hemodynamic instability,
- known hypersensitivity to any component of the oXiris set.
PRISMAFLEX
Intended use
The PRISMAFLEX control unit is intended for:
- Continuous Renal Replacement Therapy for patients with acute renal failure and/or fluid overload.
- Therapeutic Plasma Exchange Therapy for patients with diseases where removal of plasma components is indicated.
- Hemoperfusion for patients with conditions where immediate removal of substances by adsorption is indicated.
All treatments administered via the PRISMAFLEX control unit must be prescribed by a physician.
Contraindications
There are no known contraindications to Continuous Renal Replacement Therapies.
There are no known contraindications to Therapeutic Plasma Exchange.
There are no known contraindications to Hemoperfusion.
For contraindications that may apply to the disposable set selected for the therapy, refer to the Instructions For Use of the disposable set.
PRISMAFLEX HAEMODIALYSIS SETS WITH FILTER
Intended Purpose
The Prismaflex ST set is a single use device that provides blood purification through a semipermeable membrane.
The Prismaflex ST set is for use only in conjunction with the PrismaFlex control unit or with the PrisMax control unit (in countries where PrisMax is cleared or registered).
All treatments administered via the Prismaflex set must be prescribed by a physician. The size, weight, state of uremia, cardiac status, and general physical condition of the patient must be carefully evaluated by the prescribing physician before each treatment.
Indications
If patients suffer from acute kidney injury and / or volume overload, the Prismaflex ST set is indicated for continuous renal replacement therapies (CRRT), in modalities such as
- Slow Continuous UltraFiltration (SCUF)
- Continuous Veno-Venous Hemofiltration (CVVH)
- Continuous Veno-Venous HemoDialysis (CVVHD)
- Continuous Veno-Venous HemoDiaFiltration (CVVHDF) to perform fluid management and reduction of uremic toxins.
The Prismaflex ST100 and ST150 set is indicated for use in patients with a body weight equal or greater than 30 kg (66lb) and Prismaflex ST60 set is indicated to patients with a body weight greater than 11kg (24lb).
Contraindications
There are no known absolute contraindications to continuous renal replacement therapies. For the following conditions a careful assessment of the individual risk/benefit ratio has to be made by the treating physician (relative contraindications):
- inability to establish vascular access,
- severe hemodynamic instability,
- known hypersensitivity to any component of the Prismaflex set.
PrisMax
CE 0123
Prismaflex/TherMax/Oxiris/TPE Sets
CE 2797
Vantive, AN69ST, GamCath, Oxiris, Prismaflex, PrisMax and TherMax are trademarks of Vantive Health LLC or its affiliates.
References
-
Baxter International Inc. Prismaflex Operator’s Manual. Version 8.XX. Deerfield (IL): Baxter Healthcare Corporation; 2015.
-
Vantive Health LLC. Oxiris Brochure. 2025. Internal Documentation.
-
Troyanov S, Cardinal J, Geadah D, et al. Solute clearances during continuous venovenous haemofiltration at various ultrafiltration flow rates using Multilflow-100 and HF1000 filters. Nephrol Dial Transplant. 2003;18:961–966.
-
Thomas M, Moriyama K, Ledebo I. Evolution of the world's first high permeability membrane. Contrib Nephrol. 2011;173:119-129.
-
Quinto BMR, Lizuka IJ, Monte JCM, et al. TNF-α depuration is a predictor of mortality in critically ill patients under continuous veno-venous hemodiafiltration treatment. Cytokine. 2015;71(2):255-260.
-
Vantive Health LLC. TPE 1000 / TPE 2000 Specification Sheet. 2025. Internal Documentation.