Therapeutic Plasma Exchange
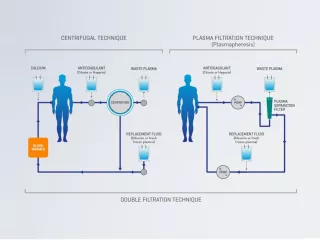
An extracorporeal blood purification technique designed for the removal of large molecular weight substances.1
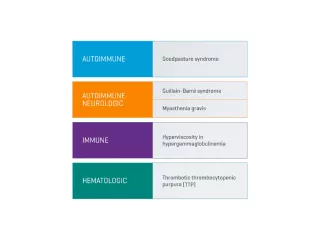
Guillain-Barré syndrome
Several controlled trials indicate that TPE can accelerate motor recovery, decrease time on the ventilator and speed of attainment of other clinical milestones compared with supportive care alone.2
Thrombotic thrombocytopenic purpura
TPE has decreased overall mortality of immune-mediated TTP from nearly uniformly fatal to <10-20%.2
Myasthenia gravis
TPE in myasthenia gravis works rapidly. Clinical effect can be apparent within 24 hours of treatment; however, in some cases it may take a week. Concomitant immunosuppression must be initiated or modified for sustained control of myasthenia gravis activity.2
Anti-glomerular basement membrane disease (Goodpasture syndrome)
A randomized controlled trial involving a small number of patients showed that TPE maintained kidney function and improved survival.2
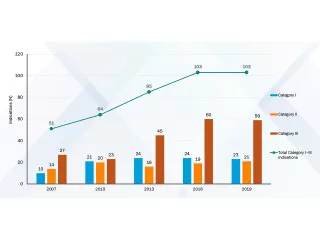
ASFA guidelines
The American Society for Apheresis (AFSA) publishes comprehensive guidelines for therapeutic apheresis. The AFSA evidence-based approach assigns diseases to categories based on a stringent review of the literature, analysis of quality of evidence and strength of recommendation derived from the evidence.
Important Safety Information
PrisMax
- The PrisMax control unit is intended for: Continuous Renal Replacement Therapy (CRRT) for patients with acute renal failure and/or fluid overload.
- Therapeutic Plasma Exchange (TPE) therapy for patients with diseases where removal of plasma components is indicated.
- Hemoperfusion(HP) for patients with conditions where immediate removal of substances by adsorption is indicated.
All treatments administered via the PrisMax control unit must be prescribed by a physician.
WARNING!
Use only fluids designed for intravenous operations and are approved for CRRT/TPE/HP usage. Do not use fluids intended for any other usage like enteral feedings/lipids or cytotoxic drugs.
CONTRAINDICATIONS
There are no known absolute contraindications to Continuous Renal Replacement Therapies.
There are no known contraindications to Therapeutic Plasma Exchange.
There are no known contraindications to Hemoperfusion.
For the following conditions a careful assessment of the individual risk/benefit ratio has to be made by the treating physician (relative contraindications).
- Inability to establish vascular access.
- Severe hemodynamic instability.
- Known hypersensitivity to any component of the Prismaflex disposable set.
TPE Sets
The Prismaflex TPE1000/TPE2000 set is indicated for use only with the PrismaFlex control unit or with the PrisMax control unit (in countries where PrisMax is cleared or registered), after having selected TPE therapy (therapeutic plasma exchange).
The Prismaflex TPE1000/TPE2000 set is intended for use in therapeutic plasma exchange, thus in diseases where removal of plasma components in indicated.
The use of the Prismaflex TPE1000 set should be restricted to patients with a body weight greater than 9kgs (20lbs).
The use of the Prismaflex TPE2000 set should be restricted to adults.
The size, weight, state of uremia, cardiac status and general physical condition of the patient must be evaluated by the prescribing physician before each treatment.
The device should be used only on the direction of a physician who has evaluated all of the pertinent features of this device in relation to the individual patient. Patients with bleeding tendencies must be closely supervised during the treatment.
Treatment should be discontinued if acute hemorrhage that can not be corrected (hematemesis, hemoptysis and melena) occurs during TPE.
All treatments administered via the Prismaflex set must be prescribed by a physician.
Contraindications
There are no known absolute contraindications to therapeutic plasma exchange.
For the following conditions a careful assessment of the individual risk/benefit ratio has to be made by the treating physician (relative contraindications):
- Inability to establish vascular access,
- Severe hemodynamic instability
- Known hypersensitivity to any component of the Prismaflex set.
For safe and proper use of the devices, please refer to the Instructions for Use.
Vantive and PrisMax are trademarks of Vantive Health LLC or its affiliates.
References
-
Kaplan AA. Therapeutic plasma exchange: core curriculum 2008. Am J Kidney Dis. 2008;52(6):1180-1196.
-
Padmanabhan A, Connelly-Smith L, Aqui N, et al. Guidelines on the use of therapeutic apheresis in clinical practice — evidence-based approach from the Writing Committee of the American Society for Apheresis: the eighth special issue. J Clin Apher. 2019;34(3):171-354.
-
Connelly-Smith L, Alquist CR, Aqui NA, et al. Guidelines on the Use of Therapeutic Apheresis in Clinical Practice - Evidence-Based Approach from the Writing Committee of the American Society for Apheresis: The Ninth Special Issue. J Clin Apher. 2023;38(2):77-278.