
PrisMax 2 System
The next-generation therapy system
The ever-changing challenges in the ICU can be demanding
At Vantive, after speaking with healthcare professionals around the world for 20+ years, we recognize the complex challenges providers face when caring for the most critically ill patients. Through this collaboration, we have identified three key challenges where Vantive can help:
1. Operational efficiency: high workload during therapy setup and delivery
2. Diverse treatment needs: patients requiring multi-organ support therapies
3. Complex clinical processes: complicated therapy setup and delivery
Compatible Products
Acute Therapy Systems
Maximize simplicity
Offer seamless therapy delivery with end-to-end intuitive interface
Simplify the complexities of CRRT and other extracorporeal therapies with the user-friendly interface of the PrisMax 2 System, which helps enable seamless therapy management, from setup to delivery.
- Large 15-inch rotating, adjustable touchscreen for multidirectional viewing of critical data even from a distance
- Simple step-by-step on-screen instructions with color coding and confirmation of completed steps with check marks
- Continuous display of vital information to simplify monitoring
- Troubleshooting information supported with images for guidance
- Smart alarms designed to reduce device interactions and treatment interruptions
Enhance EMR connectivity with TrueVue Connect for auto-documentation
Help make documentation easier with TrueVue Connect, a digital add-on communication module, which helps enable auto-documentation with enhanced EMR connectivity.
- Secure connection to the hospital’s EMR through wireless or wired connections
- TrueVue Connect communicates using HL7 and is compatible with leading EMR systems and middleware vendors
Support alkalosis risk management with the normalized Net Buffer Load (nNBL)
The normalized Net Buffer Load (nNBL) is a parameter for alkalosis risk assessment during regional citrate anticoagulation (RCA). The nNBL is continuously displayed on the user interface of the PrisMax 2 System during prescription setting and treatment.

Blood purification for sepsis management (with or without AKI)
The Prismax 2 System is compatible with the Oxiris filterset2, the only 3-in-1 blood purification filterset to effectively remove inflammatory mediators such as cytokines and endotoxins as a blood purification therapy for sepsis management. Therapy can be delivered as a stand alone hemoprofusion treatment or while delivering CRRT.2

Access TPE 24/7
With the PrisMax 2 System, you can deliver round-the-clock TPE to support time-critical indications.1
Achieve smart efficiencies
Streamline clinical workflows and increase efficiency with the PrisMax 2 System’s intelligent features that can help reduce device interactions and decrease therapy downtime.1
Optimize your CRRT program by using data-driven insights enabled by TrueVue Therapy Management.
Help optimize clinical workflows by reduced device interactions
The Auto-Effluent Drain accessory, which is available for the PrisMax 2 System, helps in optimizing the workflow in your ICU by:
- Potentially reducing bag changes by 60% and eliminating the need to change effluent bags manually, allowing more time for patient care3
- Decreasing therapy downtime and potentially increasing therapy delivery time by up to 30 minutes per patient per day3,4
- You also have the choice to capture effluent in a bag or dispose of it directly into a drain depending on the clinical situation.
Focus more on patient care with intelligent automation
Smart alarms help prevent alarm fatigue by automatically clearing alarms that are displayed for only informative purposes, such as pressure alarms. They reduce device interactions during therapy delivery enabling nursing staff to focus more on patient care.1
Advanced pressure management also minimizes alarms due to sudden pressure spikes caused by the patient moving.
Support CRRT improvements with data-driven insights
Take advantage of data-driven CRRT quality improvement programs with TrueVue Therapy Management that enables you to:
- Use your hospital-specific data to support data-driven decision-making
- Create standardized processes to help improve CRRT program efficiency
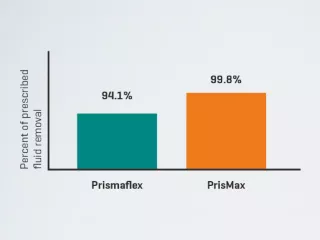
The PrisMax 2 System’s technology includes the Patient Fluid Removal (PFR) Catch-Up, which uses intelligent pump adjustments to help meet prescribed fluid removal targets. This patented technology tracks therapy delivery and safely adjusts effluent pump speed to help meet clinically prescribed fluid removal targets — even when treatment interruptions occur for up to 10 minutes per instance.1
In a clinical setting, the PrisMax 2 System delivered 99.8% of prescribed patient fluid removal.3
Important Safety Information
PrisMax
- The PrisMax control unit is intended for: Continuous Renal Replacement Therapy (CRRT) for patients with acute renal failure and/or fluid overload.
- Therapeutic Plasma Exchange (TPE) therapy for patients with diseases where removal of plasma components is indicated.
- Hemoperfusion (HP) for patients with conditions where immediate removal of substances by adsorption is indicated.
All treatments administered via the PrisMax control unit must be prescribed by a physician.
WARNING!
Use only fluids designed for intravenous operations and are approved for CRRT/TPE/HP usage. Do not use fluids intended for any other usage like enteral feedings/lipids or cytotoxic drugs.
CONTRAINDICATIONS
There are no known absolute contraindications to Continuous Renal Replacement Therapies.
There are no known contraindications to Therapeutic Plasma Exchange.
There are no known contraindications to Hemoperfusion.
Regional Citrate Anticoagulation is contraindicated in patients with severe liver failure and shock with muscle hypoperfusion.
For the following conditions a careful assessment of the individual risk/benefit ratio has to be made by the treating physician (relative contraindications).
- Inability to establish vascular access.
- Severe hemodynamic instability.
- Known hypersensitivity to any component of the Prismaflex disposable set.
GamCath
Indications
GamCath Dialysis Catheter Kits are used to obtain vascular access to perform extracorporeal blood purification. They can be used in the subclavian, jugular or femoral veins to treat patients with chronic or acute renal failure.
The GamCath Dialysis Catheter provides temporary vascular access to treat patients with chronic or acute renal failure that require extracorporeal blood purification.
Contraindications and exclusions
These catheters are not intended to be used as a treatment for existing infections.
These catheters are not intended for any use other than which is indicated.
Do not insert the catheter in thrombosed vessels.
Intended Patient Population(s)
Adult or pediatric patients except those with thrombosed vessels who are afflicted with acute or chronic renal failure and may require, at the discretion of a qualified physician, temporary vascular access for extracorporeal blood purification are suitable to be treated with GamCath Dialysis Catheter Kit.
Oxiris
Intended Purpose
The oXiris set is a single use device that provides blood purification via diffusion, convection and adsorption through a semipermeable membrane.
The oXiris set is for use only in conjunction with the PrismaFlex control unit or with the PrisMax control unit (in countries where PrisMax is cleared or registered).
All treatments administered with the oXiris set must be prescribed by a physician. The size, weight, state of uremia, cardiac status, and general physical condition of the patient must be carefully evaluated by the prescribing physician before each treatment.
Indications
The oXiris set is indicated for use in critically ill patients with a body weight equal or greater than 30 kg (66lb) for hemoperfusion and / or renal replacement modalities such as:
- Slow Continuous UltraFiltration (SCUF)
- Continuous Veno-Venous Hemofiltration (CVVH)
- Continuous Veno-Venous HemoDialysis (CVVHD)
- Continuous Veno-Venous HemoDiaFiltration (CVVHDF)
When used for hemoperfusion only, the SCUF mode shall be used with no fluid removal prescription, as the indication is to reduce elevated levels of inflammatory mediators, such as cytokines, and to reduce endotoxins.
If patients suffer from acute kidney injury and / or volume overload, the oXiris set is indicated for continuous renal replacement therapies (CRRT), to perform fluid management and removal of uremic toxins. The removal of inflammatory mediators and endotoxins is performed simultaneously when indicated for CRRT.
Contraindications
It is contraindicated to use the oXiris set where patients present a known allergy to heparin or have type II thrombocytopenia caused by heparin (HIT Syndrome type II).
Any contraindications indicated in the respective Instructions for use of all drugs used simultaneously with this set should be taken into account.
For the following conditions a careful assessment of the individual risk/benefit ratio has to be made by the treating physician (relative contraindications):
- inability to establish vascular access,
- severe hemodynamic instability,
- known hypersensitivity to any component of the oXiris set.
TherMax Blood Warmer Unit
Intended use
The TherMax Blood Warmer Accessory includes the Blood Warmer Unit and Blood Warmer Disposable. It is intended for use in combination with the PrisMax control unit using Prismaflex blood tubing sets for the warming of blood during extracorporeal blood circulation
Indications for use
The TherMax Blood Warmer Accessory is indicated for warming returning blood flow.
Contraindications
There are no known contraindications when used as intended, and within the specified operating conditions.
PRISMAFLEX
Intended use
The PRISMAFLEX control unit is intended for:
- Continuous Renal Replacement Therapy for patients with acute renal failure and/or fluid overload.
- Therapeutic Plasma Exchange Therapy for patients with diseases where removal of plasma components is indicated.
- Hemoperfusion for patients with conditions where immediate removal of substances by adsorption is indicated.
All treatments administered via the PRISMAFLEX control unit must be prescribed by a physician.
Contraindications
There are no known contraindications to Continuous Renal Replacement
Therapies.
There are no known contraindications to Therapeutic Plasma Exchange.
There are no known contraindications to Hemoperfusion.
For contraindications that may apply to the disposable set selected for the therapy, refer to the Instructions For Use of the disposable set.
TrueVue Connect is a communications module designed to add additional communications and networking functionality to a PrisMax device.
For safe and proper use of the devices, please refer to the Instructions for Use.
CE 0123
CE 2797
Vantive, AN69ST, GamCath, Oxiris, Prismaflex, PrisMax, TherMax and TrueVue are trademarks of Vantive Health LLC or its affiliates.
References
-
Vantive Health LLC. PrisMax Operator’s Manual. 2020. AW8046.
-
Vantive Health LLC. Oxiris Market Research 3-in-1. 2021. Internal Documentation.
-
Vantive Health LLC. PrisMax Limited Controlled Distribution Report. 2018. Internal Documentation.
-
Vantive Health LLC. Statistical Analysis, Summary of PrisMax LCD Data. 2018. Internal Documentation.
-
Vantive Health LLC. TherMax Operator’s Manual. 2018. AW7006.



