Oxiris Set
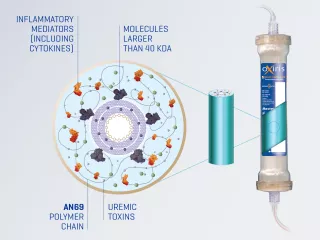
Oxiris is the only 3-in-1 blood purification filterset to effectively remove inflammatory mediators such as cytokines and endotoxins as a blood purification therapy for sepsis management. Therapy can be delivered as a stand alone hemoprofusion treatment or while delivering CRRT.1-5
Sepsis is a major and growing global healthcare challenge.6-8 Blood purification therapies may help improve outcomes.1,3,9-11
~30% of ICU patients have sepsis12
~50% of patients with sepsis have kidney dysfunction or failure13,14
~45% of patients with sepsis die within 1 year7,8,15
Compatible Products
Acute Therapy Systems
Filter Sets & Catheters
Digital Health
Versatile innovation
The Oxiris set can be used to deliver hemoperfusion to reduce elevated levels of inflammatory mediators/endotoxins and/or to deliver any CRRT modality (SCUF, CVVH, CVVHD, CVVHDF) to provide uremic toxin removal and support fluid management.5 Note: CVVH, Continuous Veno-Venous Hemofiltration; CVVHD, Continuous Veno-Venous Hemodialysis; CVVHDF, Continuous Veno-Venous Hemodiafiltration; SCUF, Slow Continuous UltraFiltration.
Inflammatory mediator removal
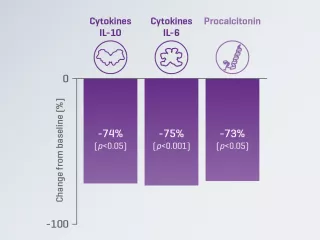
A prospective, multicenter, observational study reported a significant decrease from baseline in serum IL-6 levels in patients with COVID-19 treated with the Oxiris set.2
Endotoxin removal
A randomized, crossover study reported significantly greater changes from baseline in endotoxin level with the Oxiris set, compared to the AN69ST filterset in patients with septic-shock-associated AKI.3
Versatile simplification
The Oxiris filterset is designed to simplify blood purification therapy delivery for patients requiring both cytokine/endotoxin removal and CRRT, which may free up nursing time. The Oxiris set is part of a pre-connected set for simplicity. The closed system includes a filter membrane pre-connected to the color-coded tubing, minimizing setup steps and reducing potential for touch contamination.5,16
The Oxiris set can play a role in the management of patients with COVID-19
In a prospective observational pilot study of COVID-19 patients (n=37) admitted to an ICU in Italy between February and April 2020, patients were treated with extracorporeal blood purification (EBP) using the Oxiris set for immunomodulation and/or support to renal function during AKI.2 Results suggest that early initiation of EBP during COVID-19 may be associated with greater benefits.
87%: IL-6 serum levels decrease during the first 72 hours of treatment vs. baseline (p < 0.001)*
4.5: Median SOFA score decrease† from 13 at baseline to 8.5 after 72 hours of treatment (p = 0.002)‡
8.3%: Lower observed mortality rate vs. expected mortality rate§ of historical control (based on APACHE IV¶ score)
*IL-6 reduction was statistically significant over the first 72 hours of treatment (p<0.001), and for each time period (24h, 48h, 72h) compared to baseline (p=0.001). p<0.016 considered for statistical significance.
†Sequential Organ Failure Assessment.
‡SOFA score reduction was statistically significant over the first 72 hours of treatment (p=0.002), and at 48h vs baseline (p=0.001) and 72h vs baseline (p=0.002). p<0.016 considered for statistical significance.
§Due to the observational design of this study (single arm without active control group), it was not possible to determine whether a causal relationship exists between EBP with Oxiris and improvement in patient outcomes (organ function or mortality). Given the small number of patients studied, it was not possible to identify any parameter values or illness stage that might indicate a threshold or cutoff for the best time to initiate EBP.
¶Acute Physiological and Chronic Health Evaluation (APACHE) is a scoring system for disease severity that uses actual clinical data obtained on the first day of ICU admission to estimate the risk of short-term mortality as well as predict the length of ICU stay.
Explore the clinical data
Evidence suggests that the Oxiris set is associated with clinical improvement in organ function (as measured by SOFA score) and hemodynamic parameters.1,3,20,21
The COVID-19 pandemic
Systemic inflammation in patients with COVID-19 can lead to multi-organ failure, including AKI.22,23
Blood purification techniques have been shown to remove cytokines, endotoxin and circulating viral particles in patients with COVID-19.24
Blood purification is the only therapy that may effectively remove inflammatory mediators, damage-associated molecular patterns (DAMPs) and/or pathogen-associated molecular patterns (PAMPs, e.g., endotoxin),2 which may help mitigate progression to multi-organ failure.1,2,9,11,20,21,25
Potential targets for COVID-19 therapies include the removal of cytokines26
Stages of the SARS-CoV-2 life cycle offer potential treatment targets. Potential targets for COVID-19 therapies include anti-inflammatory therapies,27 prevention of membrane fusion and viral cell entry,28 removal of cytokines or prevention of cytokine receptor activation26,28,29 and prevention of RNA translation.28,30-33
Important Safety Information
Oxiris
Intended Purpose
The oXiris set is a single use device that provides blood purification via diffusion, convection and adsorption through a semipermeable membrane.
The oXiris set is for use only in conjunction with the PrismaFlex control unit or with the PrisMax control unit (in countries where PrisMax is cleared or registered).
All treatments administered with the oXiris set must be prescribed by a physician. The size, weight, state of uremia, cardiac status, and general physical condition of the patient must be carefully evaluated by the prescribing physician before each treatment.
Indications
The oXiris set is indicated for use in critically ill patients with a body weight equal or greater than 30 kg (66lb) for hemoperfusion and / or renal replacement modalities such as:
- Slow Continuous UltraFiltration (SCUF)
- Continuous Veno-Venous Hemofiltration (CVVH)
- Continuous Veno-Venous HemoDialysis (CVVHD)
- Continuous Veno-Venous HemoDiaFiltration (CVVHDF)
When used for hemoperfusion only, the SCUF mode shall be used with no fluid removal prescription, as the indication is to reduce elevated levels of inflammatory mediators, such as cytokines, and to reduce endotoxins.
If patients suffer from acute kidney injury and / or volume overload, the oXiris set is indicated for continuous renal replacement therapies (CRRT), to perform fluid management and removal of uremic toxins. The removal of inflammatory mediators and endotoxins is performed simultaneously when indicated for CRRT.
Contraindications
It is contraindicated to use the oXiris set where patients present a known allergy to heparin or have type II thrombocytopenia caused by heparin (HIT Syndrome type II).
Any contraindications indicated in the respective Instructions for use of all drugs used simultaneously with this set should be taken into account.
For the following conditions a careful assessment of the individual risk/benefit ratio has to be made by the treating physician (relative contraindications):
- inability to establish vascular access,
- severe hemodynamic instability,
- known hypersensitivity to any component of the oXiris set.
PRISMAFLEX HEMOFILTER SETS
Intended Purpose
The Prismaflex ST set is a single use device that provides blood purification through a semipermeable membrane.
The Prismaflex ST set is for use only in conjunction with the PrismaFlex control unit or with the PrisMax control unit (in countries where PrisMax is cleared or registered).
All treatments administered via the Prismaflex set must be prescribed by a physician. The size, weight, state of uremia, cardiac status, and general physical condition of the patient must be carefully evaluated by the prescribing physician before each treatment.
Indications
If patients suffer from acute kidney injury and / or volume overload, the Prismaflex ST set is indicated for continuous renal replacement therapies (CRRT), in modalities such as
- Slow Continuous UltraFiltration (SCUF)
- Continuous Veno-Venous Hemofiltration (CVVH)
- Continuous Veno-Venous HemoDialysis (CVVHD)
- Continuous Veno-Venous HemoDiaFiltration (CVVHDF) to perform fluid management and reduction of uremic toxins.
The Prismaflex ST100 and ST150 set is indicated for use in patients with a body weight equal or greater than 30 kg (66lb) and Prismaflex ST60 set is indicated to patients with a body weight greater than 11kg (24lb).
Contraindications
There are no known absolute contraindications to continuous renal replacement therapies. For the following conditions a careful assessment of the individual risk/benefit ratio has to be made by the treating physician (relative contraindications):
- inability to establish vascular access,
- severe hemodynamic instability,
- known hypersensitivity to any component of the Prismaflex set.
For safe and proper use of the devices, please refer to the Instructions for Use.
CE 0123
CE 2797
Vantive, AN69ST, Oxiris, Prismaflex and PrisMax are trademarks of Vantive Health LLC or its affiliates.
References
-
Turani F, Barchetta R, Falco M, Busatti S, Weltert L. Continuous renal replacement therapy with the adsorbing filter Oxiris in septic patients: a case series. Blood Purif. 2019;47(3):54-58.
-
Villa G, Romagnoli S, De Rosa S, et al. Blood purification therapy with a hemodiafilter featuring enhanced adsorptive properties for cytokine removal in patients presenting COVID-19: a pilot study. Crit Care. 2020;24:605.
-
Broman ME, Hansson F, Vincent JL, Bodelsson M. Endotoxin and cytokine reducing properties of the oXiris membrane in patients with septic shock: a randomized crossover double-blind study. PLoS One. 2019;14:e0220444.
-
Malard B, Lambert C, Kellum JA. In vitro comparison of the adsorption of inflammatory mediators by blood purification devices. Intensive Care Med Exp. 2018;6:12.
-
Vantive Health LLC. Oxiris Instructions for Use. 2022.
-
Rudd KE, Johnson SC, Agesa KM, et al. Global, regional, and national sepsis incidence and mortality, 1990-2017: analysis for the Global Burden of Disease Study. Lancet. 2020;395:200-211.
-
Karlsson S, Varpula M, Ruokonen E, et al. Incidence, treatment, and outcome of severe sepsis in ICU-treated adults in Finland: the Finnsepsis study. Intensive care medicine. 33. 435-43. 10.1007/s00134-006-0504-z. et al. Intensive Care Med. 2007;33:435-443.
-
van Vught LA, Klein Klouwenberg PM, Spitoni C, et al. Incidence, risk factors, and attributable mortality of secondary infections in the intensive care unit after admission for sepsis. JAMA. 2016;315:1469-1479.
-
Lumlertgul N, Srisawat N. The haemodynamic effects of oXiris haemofilter in septic shock patients requiring renal support: A single-centre experience. Int J Artif Organs. 2020;44:17-24.
-
Cruz DN, Antonelli M, Fumagalli R, et al. Early use of polymyxin B hemoperfusion in abdominal septic shock: the EUPHAS randomized controlled trial. JAMA. 2009;301:2445-2452.
-
Hawchar F, László I, Öveges N, Trásy D, Ondrik Z, Molnar Z. Extracorporeal cytokine adsorption in septic shock: a proof of concept randomized, controlled pilot study. J Crit Care. 2019;49:172-178.
-
Sakr Y, Jaschinski U, Wittebole X, et al. Sepsis in intensive care unit patients: worldwide data from the Intensive Care over Nations Audit. Open Forum Infect Dis. 2018;5:ofy313.
-
Yébenes JC, Ruiz-Rodriguez JC, Ferrer R, et al. Epidemiology of sepsis in Catalonia: analysis of incidence and outcomes in a European setting. Ann Intensive Care. 2017;7:19.
-
Mayr FB, Yende S, Linde-Zwirble WT, et al. Infection rate and acute organ dysfunction risk as explanations for racial differences in severe sepsis. JAMA. 2010;303:2495-2503.
-
Prescott HC, Osterholzer JJ, Langa KM, Angus DC, Iwashyna TJ. Late mortality after sepsis: propensity matched cohort study. BMJ. 2016;353:i2375.
-
Vantive Health LLC. Oxiris Specification Sheet. 2022.
-
Monard C, Rimmelé T, Ronco C. Extracorporeal blood purification therapies for sepsis. Blood Purif. 2019;47(Suppl. 3):2-15.
-
Thomas M, Moriyama K, Ledebo I. AN69: Evolution of the world's first high permeability membrane. Contrib Nephrol. 2011;173:119-129.
-
Hattori N, Oda S. Cytokine-adsorbing hemofilter: old but new modality for septic acute kidney injury. Ren Repl Ther. 2016;2:41.
-
Schwindenhammer V, Girardot T, Chaulier K, et al. oXiris® use in septic shock: experience of two French centres. Blood Purif. 2019;47(suppl 3):29-35.
-
Nassiri AA, Hakemi MS, Miri MM, Shahrami R, Koomleh AA, Sabaghian T. Blood purification with CytoSorb in critically ill COVID-19 patients: a case series of 26 patients. Artif Organs. 2021;45:1338-1347.
-
Lavillegrand JR, Garnier M, Spaeth A, et al. Elevated plasma IL-6 and CRP levels are associated with adverse clinical outcomes and death in critically ill SARS-CoV-2 patients: inflammatory response of SARS-CoV-2 patients. Ann Intensive Care. 2021;11:9.
-
Anderberg S, Luther T, Berglund M, et al. Increased levels of plasma cytokines and correlations to organ failure and 30-day mortality in critically ill Covid-19 patients. Cytokine. 2021;138:155389.
-
Nadim MK, Forni LG, Mehta RL, et al. COVID-19-associated acute kidney injury: consensus report of the 25th Acute Disease Quality Initiative (ADQI) Workgroup. Nat Rev Nephrol. 2020;16:747-764.
-
Rosalia RA, Ugurov P, Neziri D, et al. Extracorporeal blood purification in moderate and severe COVID-19 patients: a prospective cohort study. Blood Purif. 2021;51:233-242.
-
Ronco C, Bagshaw SM, Bellomo R, et al. Extracorporeal blood purification and organ support in the critically ill patient during COVID-19 pandemic: expert review and recommendation. Blood Purif. 2021;50(1):17-27.
-
RECOVERY Collaborative Group, Horby P, Lim WS, et al. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384(8):693-704.
-
Sanders JM, Monogue ML, Jodlowski TZ, Cutrell JB. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): a review. JAMA. 2020;323(18):1824-1836.
-
Huet T, Beaussier H, Voisin O, et al. Anakinra for severe forms of COVID-19: a cohort study. Lancet Rheumatol. 2020;2:e393-400.
-
Hung IF, Lung KC, Tso EY, et al. Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial. Lancet. 2020;395:1695-704.
-
Sang P, Tia S, Meng Z, Yang L. Anti-HIV drug repurposing against SARS-CoV-2. RSC Adv. 2020;10:15775-15783.
-
Beigel JH, Tomashek KM, Dodd LE, et al. Remdesivir for the treatment of Covid-19 - final report. N Engl J Med. 2020;383(19):1813-1826.
-
U.S. National Library of Medicine. NCT04351295. Available at: https://clinicaltrials.gov/ct2/show/NCT04351295 (accessed November 2021).